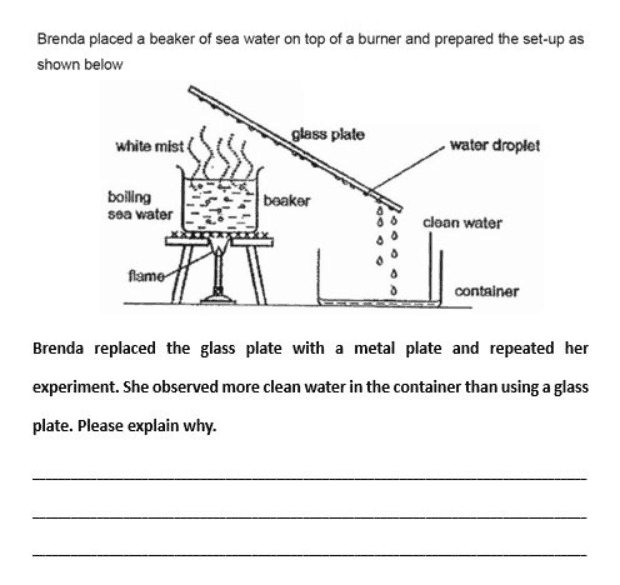
My child said this is because the metal plate loses heat to the enviornment air faster, while I think this is because the metal plate gains heat from the water vapor faster. Need some advice. Thanks in advance!


My child said this is because the metal plate loses heat to the enviornment air faster, while I think this is because the metal plate gains heat from the water vapor faster. Need some advice. Thanks in advance!
Both of your observations are correct but your son’s / daughter’s observation is more relevant in terms of answering the question:
“… the metal plate loses heat to the environment air faster“
I understand why your child said this and he/she is not wrong. Metal is a better conductor of heat and this means that it will both transfer and absorb heat at a faster rate. When the metal plate transfers (loses) heat to the environment air, it will cool and allow the process of condensation to quicken.
The glass plate however, will heat up more slowly compared to the metal plate, since it is not a good conductor of heat. But it will retain the heat and maintain its hotness for a much longer time as well since it cannot transfer the heat away as readily as the metal plate. This can slow down condensation.
“… the metal plate gains heat from the water vapor faster“
Again, this is not wrong but it does not really answer the question. The metal plate will indeed absorb (gain) heat from the water vapour faster but will also lose it faster relative to the glass plate.
For a Primary level question, my suggested answer would be:
The metal plate is a better conductor of heat and conducts heat away from the steam at a faster rate. As the steam loses heat to the metal plate faster, the process of condensation will quicken and more water droplets will form on the metal plate, allowing for more clean water to be collected.
Wow! Thanks a lot for your detailed explanation and precise answer!